Hairmax® Blog

The Laser Advantage: Why Lasers Prevail Over LEDs for Effective Hair Growth
You may see advertisements about LED hair growth devices. Don’t be fooled by companies that claim hair growth results from using LEDs instead of Lasers. There are significant differences between...
Read More

Hairmax Vs. iRestore: Why Hairmax is the Superior Choice for Laser Hair Growth Therapy
Exploring your options for combating hair loss? Laser Therapy emerges as a beacon of hope, especially with FDA-cleared, side-effect-free solutions available for both men and women. Amidst the myriad of...
Read More

Revolutionizing Personal Investment: Exploring the Science Behind Innovative Hair Growth Solutions
In the realm of personal investment, where the pursuit of financial growth meets the quest for innovation, a fascinating transformation is taking place.
Read More

Does Our Hair Age Too?
Today, we are talking about an important topic - the aging of our hair. It is no secret that our hair ages right along with us. Look at the photos...
Read More

Hair Loss: How To Determine and Identify It's Stages
Hair loss is a concern that affects many men and women. Recognizing the presence of hair loss and understanding its stages
Read More

Trichologist Talks About Her Mistake and Laser Hair Treatment
During my consultations after the end of the diagnosis with the help of trichoscopy or phototrichogram and the determination of the causes of hair loss, I proceed to prescribe the...
Read More
Hair Loss Expert - Trichologist Explains Androgenetic alopecia
Androgenetic alopecia is one of the most common types of hair loss, but as my personal experience shows, patients know very little about it.
Read More

Traction Alopecia: Is your hairstyle contributing to your hair loss?
I had an interesting case at the beginning of my career as a trichologist. I was working as the chief physician in a large clinic in Baku, Azerbaijan and
Read More

Effective Prevention Tips and Remedies for Postpartum Hair Loss
Postpartum alopecia is one of the conditions that may affect women in the months following childbirth. New mothers lose about 400 hairs a day,
Read More

Hairmax Welcomes Trichologist, Shab Caspara as Brand Ambassador
Trichologist and Hair Expert, Shab Caspara, has joined Hairmax, the laser hair growth leader, as a Brand Ambassador.
Read More
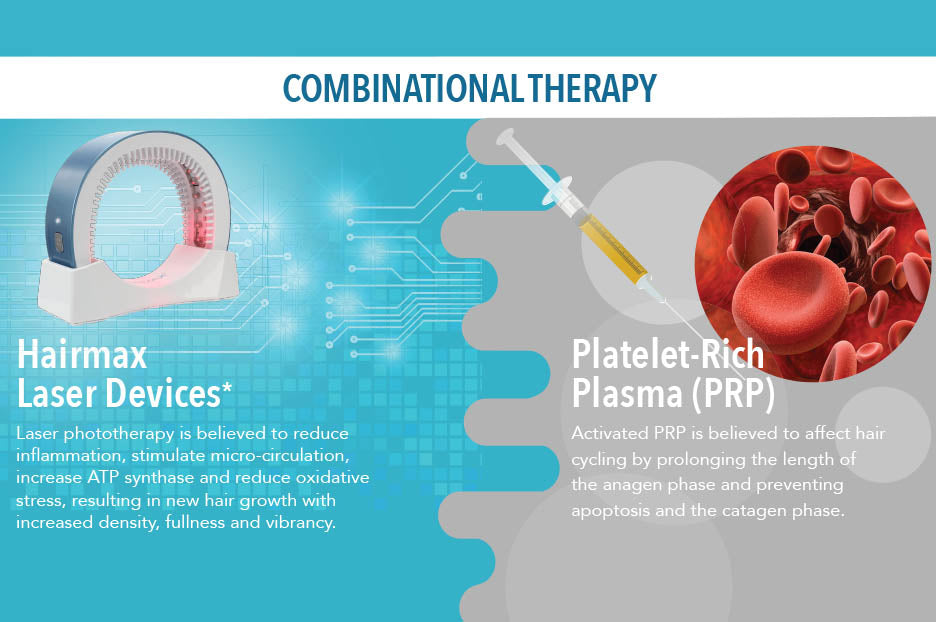
Hairmax and PRP Together for Enhanced Results
Hairmax laser therapy and PRP therapy - a winning combination of techniques in the fight against hair loss. One of the main trends of modern life is a healthy lifestyle....
Read More

Hairmax welcomes Yuliia Chorna, Ukrainian Dermatologist as Expert Advisor
Boca Raton, Florida – Yuliia Chorna, Certified Trichologist IAT, WTS, and Dermatologist with over 23 years of experience in hair loss and scalp disorders, has joined Hairmax, the leader in...
Read More

Regrow Your Hair! Tips to Regain Self-Confidence After Cancer Treatment
Hair loss is probably one of cancer treatment’s most distressing side effects. For instance, chemotherapy which targets cancer cells may also damage healthy cells responsible for hair growth.
Read More

Don't Let Long Flight Ruin Your Look - Follow These 7 Tips to Care for Your Hair
Have you ever noticed how much damage air travel can do to your hair? Planes rely on recycled dry air, and with high altitude combined with static-prone materials like nylon...
Read More

6 Myths About Laser Hair Treatment
Laser hair treatments can offer an easy way to stimulate hair growth at home. If you've noticed thinning hair or slowly developing bald spots, we suggest taking action quickly to...
Read More

10 Hair Care Tips For Winter
The weather outside may be frightful, but there are steps you can take to help protect your hair from cold weather trauma. Cold air can make your scalp dry and...
Read More

ENTER NOW: $2500 Live Your Best Life Giveaway Sweepstakes, features Hairmax PLUS 5 other Luxury Brands.
We’ve teamed up with FindKeep.Love and some of the most of-the-moment brands in beauty, fashion, and more for a VIP sweepstakes.
Read More

POST-CHEMOTHERAPY HAIR LOSS AND WHAT YOU DO ABOUT IT
Being diagnosed and treated for hair loss after chemotherapy, but also losing your hair, Hairmax may be a good way to help restore your hair and your self-esteem...
Read More






















What About Watts? HairMax Laser Technology explained!
Medical grade lasers - not LEDs - deliver the optimal amount of light energy to stimulate hair growth and reverse the thinning process. HairMax lasers produce collimated light, which delivers...
Read More



Dr. Cabral Explains Why The HairMax LaserBand 82 Works!
Dr. Stephen Cabral gives his honest product review of the LaserBand 82, shared on the Cabral Concept podcast. Dr. Cabral has appeared in every type of media outlet as a...
Read More

Get Inspired! Start Living Your Life Today
It's time to get inspired with Jason Rosell. You've seen him on the Today Show, Hallmark Channel, The Steve Harvey Show and more. Jason is here to tell you how...
Read More

Dr. Gadget's Must-Haves: HairMax Powerflex Cap and LaserBand 82
We're happy to be featured in Dr. Gadget's segment on Fox LA. The HairMax LaserBand 82 and PowerFlex 272 Laser Cap are not really gadgets, but FDA Cleared Medical Devices...
Read More

Red Light Your Way To Healthy Hair!
The HairMax LaserBand 82 is featured in a segment on Red Light Therapy on Access Online nationwide. Learn more about HairMax and how Red Laser Light Therapy can help regrow...
Read More

Bio-Active Thinning Hair Care Explained
What is Bio-Active Hair Therapy? How does it work to help support healthy hair? This and more explained by Randy Veliky, Chief Technology Officer and Francesca Dubsky, Director of ...
Read More

The Top 5 questions about Hair Loss and HairMax Answered!
A new series by HairMax - Your top 5 questions about Hair Loss and HairMax answered! Everything you always wanted to know, but didn't know who to ask... until now. Hosted...
Read More

Huda Kattan opens up about her struggle with balding and hair loss
Huda Kattan is opening up about a beauty woe that's plagued her for years: hair loss. In a raw new video, the makeup mogul reveals that she's dealt with balding and...
Read More

SAG Awards 2020: See What's Inside This Year's Gift Bags
Last night saw the 2020 Screen Actors Guild awards, held this year at LA’s Shrine Auditorium and Expo Hall. And with nominees from Jodie Comer and Phoebe Waller-Bridge to Charlize...
Read More






